Cold Chain Solutions
Introtech is known as the cold chain specialist.
We offer measurement solutions for every phase in the cold chain.
Each solution enables your organization to proof regulatory compliance.
- Transport monitoring to document deviations in critical conditions;
- Temperature mapping to proof that your facility is fit for it’s intended use;
- Central monitoring to secure and safeguard your facilities continuously.
Solutions throughout the entire Cold Chain.

Transport monitoring
According to the e-GDP regulations organizations need to ensure that the consignments are correct, that the products originate from approved suppliers and that they have not been visibly damaged during transport.
IntroTech offers easy to use solutions to document deviations in temperature, humidity, shocks and location to enable your organization to proof regulatory compliance.
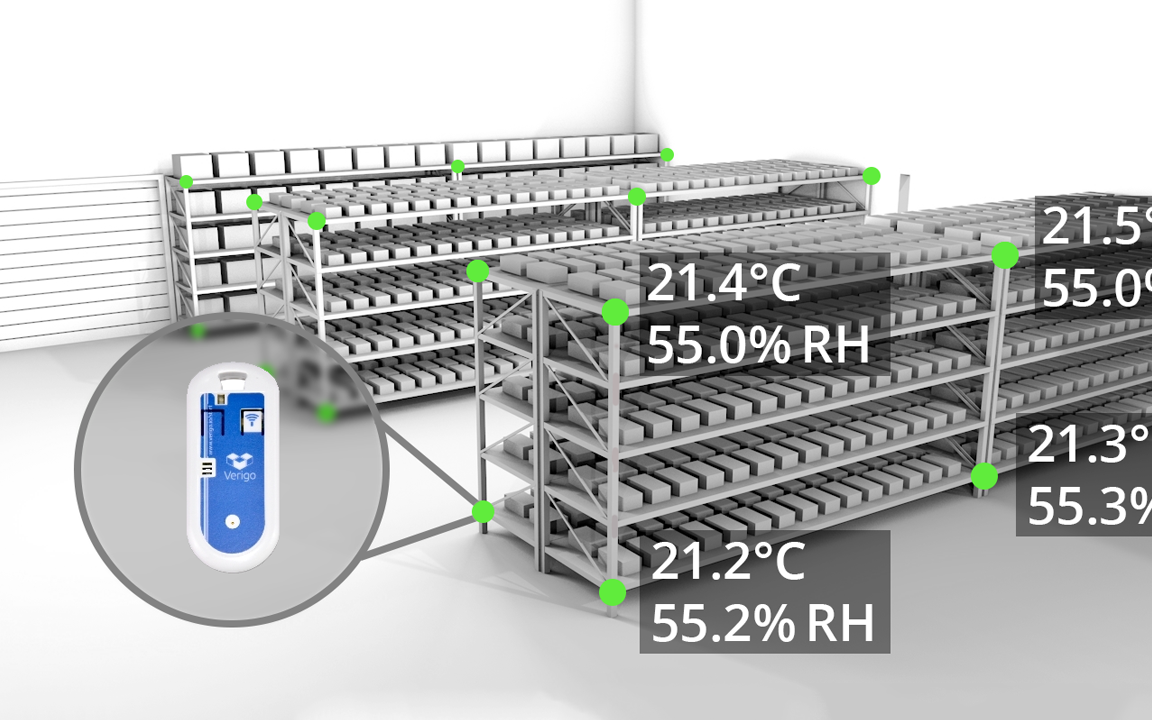
Temperature mapping
Temperature mapping is a requirement in the context of e-GDP/e-GMP regulations for each space intended for the storage and handling of products with a specific allocated storage temperature and humidity specification.
Determine the hot and cold spots of an area to allow the installation of sensor for permanent registration. IntroTech can provide the necessary hard- and software, or perform the entire mapping process.
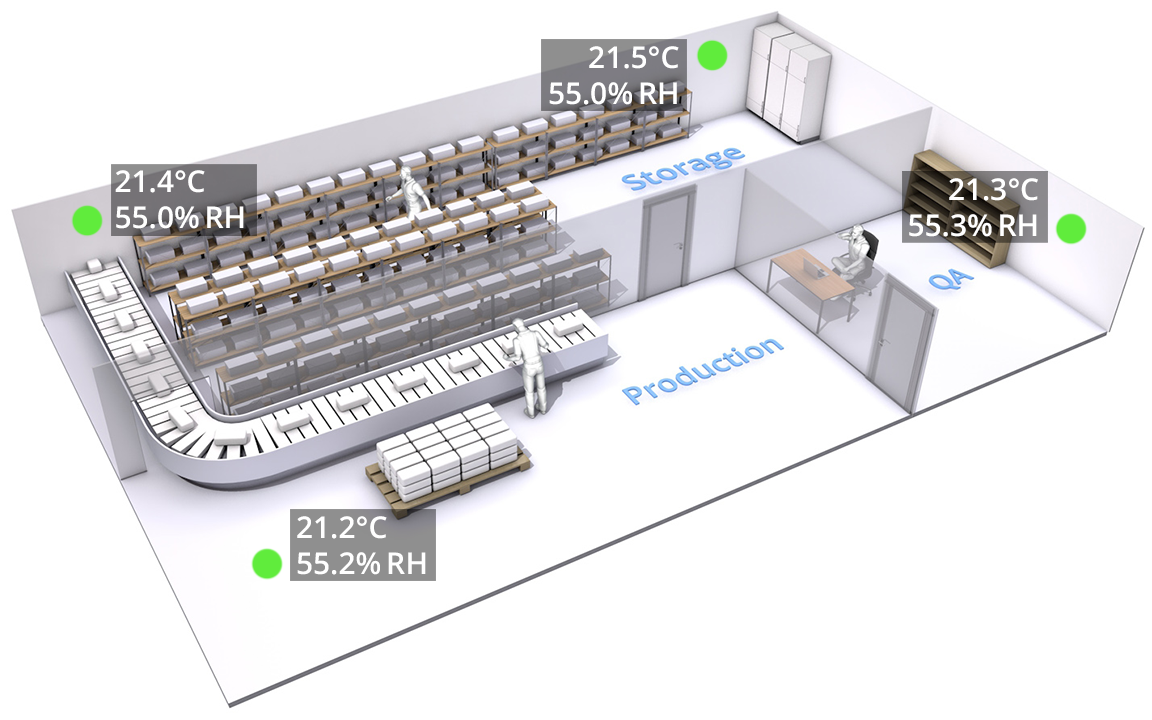
Central monitoring
The e-GDP/e-GMP regulations for the pharmaceutical industry prescribe that once the critical control points are located by temperature mapping, each control point should be monitored 24/7.
In addition, regulations demand that the organization is notified whenever a critical point exceed limits. IntroTech can provide the required hard- and software, or help implement monitoring solutions.